John-William Sidhom
Hematology & Medical Oncology Fellow
Weill Cornell Medicine
NewYork-Presbyterian Hospital
Welcome!
Thanks for stopping by and checking out my personal page. I am a clinical fellow in hematology & medical oncology at Weill Cornell Medicine, NewYork-Presbyterian Hospital. Prior to joining Weill Cornell for fellowship, I completed an MD/PhD from the Johns Hopkins University within the School of Medicine and Whiting School of Engineering, followed by my internal medicine residency at the Mount Sinai Hospital in the Icahn School of Medicine.
Current Work
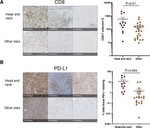
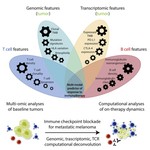
At Weill Cornell, I am co-mentored by Dr. Manish A. Shah (Chief, Solid Tumor Service) and Dr. Olivier Elemento (Director, Englander Institute for Precision Medicine). My research develops AI foundation models for the cancer genome: large-scale deep learning systems trained on tumor sequencing data, designed to guide treatment selection in solid tumors. Current applications focus on colorectal and pancreatic cancer. This work is supported by the 2026 ASCO Young Investigator Award and a Weill Cornell × Mastercard AI / Big Data Seed Grant, and has been presented at the 2025 AACR Annual Meeting (Scholar-in-Training Award). My clinical interest is in GI oncology.
Doctoral Work
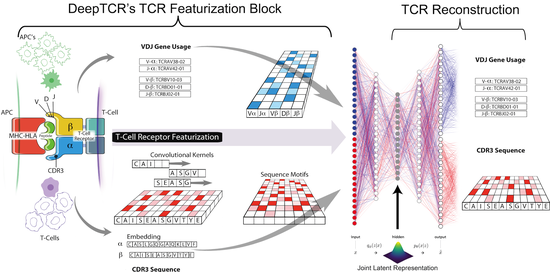
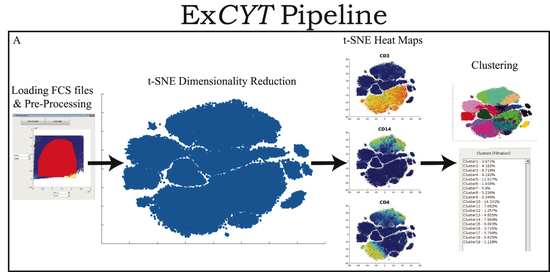
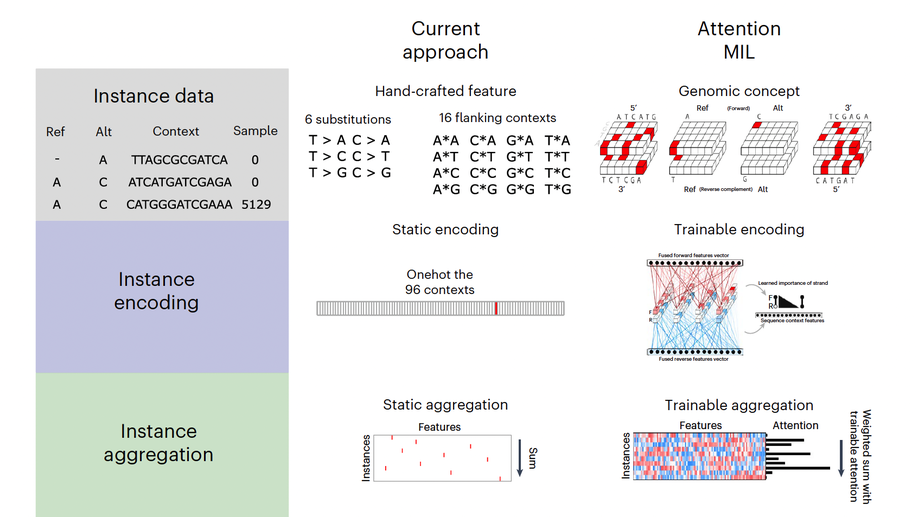
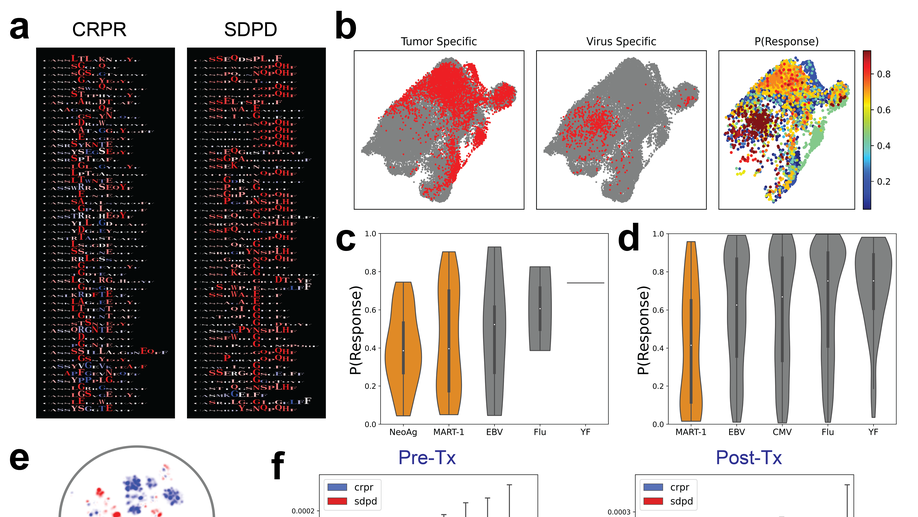
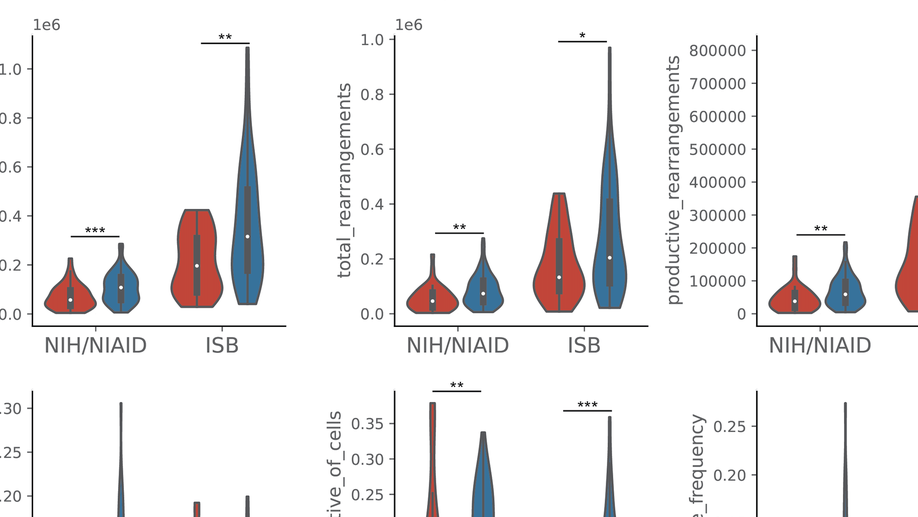
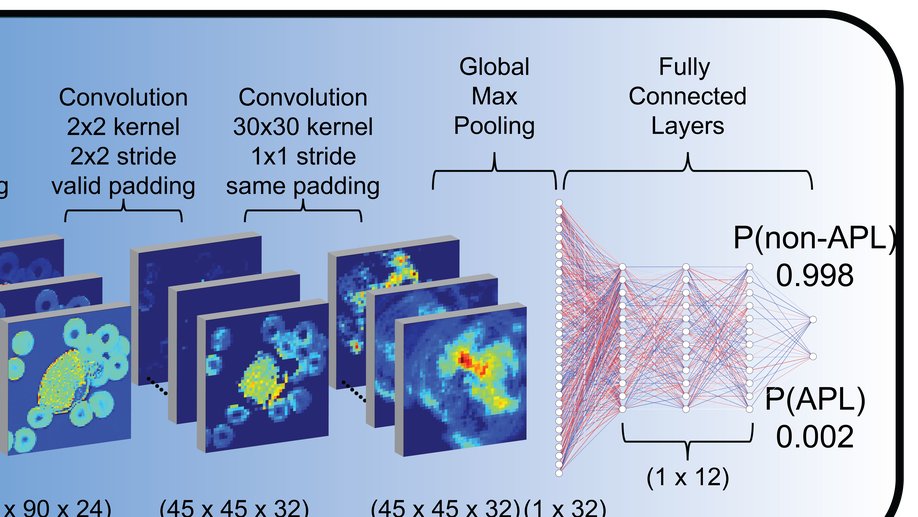
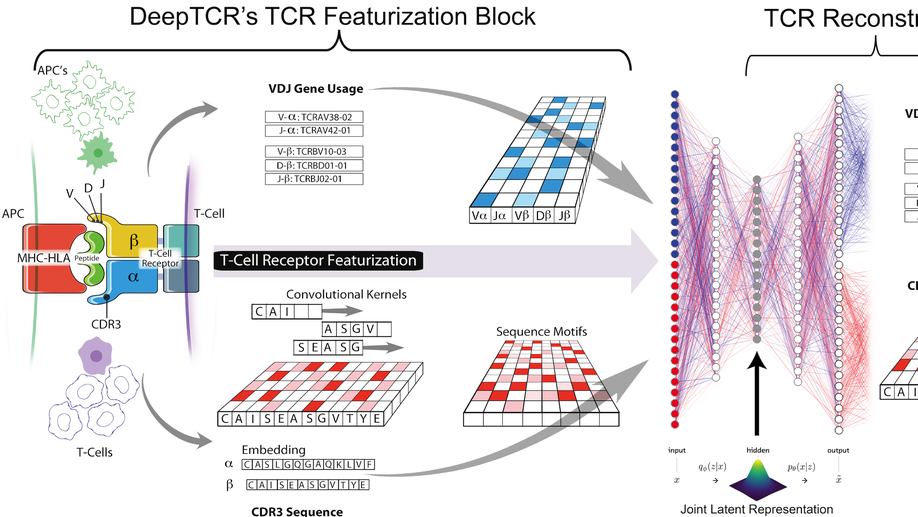
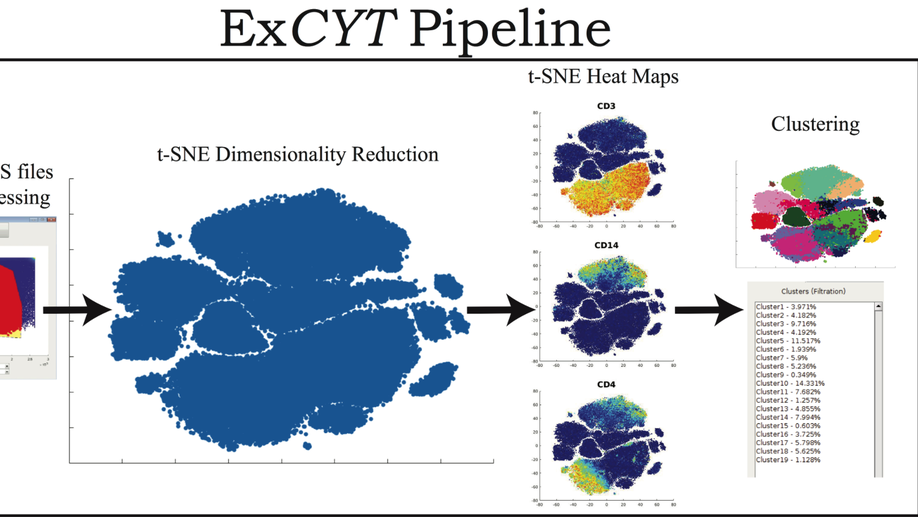
My doctoral work (Thesis Advisor: Drew M. Pardoll, MD., PhD., Co-Mentor: Alexander S. Baras, MD., PhD.) was at the intersection of artificial intelligence, machine learning, and cancer immunogenomics, where I developed algorithmic approaches such as DeepTCR for T-cell repertoire analysis and the ATGC framework for somatic mutation modeling to study the interaction of cancer and the immune system.
Pre-Doctoral & Master’s Work
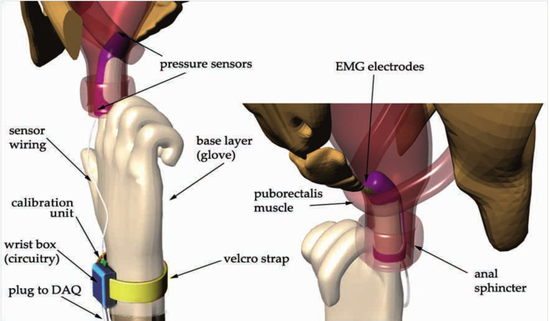
Before the MD/PhD, I completed a Bachelor of Science in Engineering in Biomedical Engineering with a minor in mathematics and certificate of entrepreneurship at the University of Michigan in 2011, followed by a Master’s of Science & Engineering in Biomedical Engineering at the Johns Hopkins Center for Bioengineering Innovation and Design (CBID experience here). My focus was medical device design, including the CryoPop, a low-cost cryotherapy unit for the treatment of cervical precancer. CryoPop has been patented, completed its first clinical trial (NCT02367625), is enrolling in a second (NCT04154644), and is commercially available through Pregna International Ltd.
Hobbies & Interests
Outside of the hospital and the lab, I am a competitive Olympic weightlifter (American Open Series qualifier, 2017) and an endurance athlete (multiple half-marathons, Olympic-distance triathlon, Ironman 70.3, and long-distance charity swims with Swim Across America). I also enjoy producing music (piano, saxophone, guitar, vocals) and amateur photography. Otherwise, I enjoy visiting new cities, eating good food, and spending time with friends, family, and my dog Leo.
Contact Me!
I hope this page provides insight into the work I have done and areas of research I hope to continue to pursue in my future career as well as my own personal interests! Feel free to contact me and reach out with any questions or research collaboration opportunities!
Interests
- Cancer Immunology
- Artificial Intelligence
- Deep Learning
- Large Language Models
- Immunogenomics
- Precision Oncology
- Gastrointestinal Oncology
Education
Doctorate of Medicine, 2021
Johns Hopkins University School of Medicine
Doctorate of Philosophy in Biomedical Engineering, 2021
Johns Hopkins University Whiting School of Engineering
Master's of Science & Engineering in Biomedical Engineering, 2012
Johns Hopkins University Whiting School of Engineering
Bachelor of Science in Engineering in Biomedical Engineering (summa cum laude), 2011
University of Michigan
Deep Learning Nanodegree
Udacity School of AI